Labeling High and Low Energy Surfaces

There are many factors which affect how well a label is going to stick to a specific material surface such as how textured it is, the porosity, is the surface coated e.g. paint, lacquer, grease, etc. and crucially surface energy. In the world of labeling, surface energy relates to how well a label adhesive is going to stick to a specific application surface.
What is surface energy?
In technical terms surface energy is defined as the sum of all intermolecular forces that are on the surface of a material. More simply, surface energy is the degree to which the adhesive and the material's surface repel or attract. Surface tension is normally measured in energy units called dynes/cm. A dyne is the amount of force required to produce an acceleration of 1 cm/sec²on a mass of 1g. The dyne level of a material is called its surface energy.
Low Surface Energy (LSE)
The lower the surface energy of an application material the harder adhesion becomes, resulting in labels peeling or falling off altogether. Typical low surface materials include; PVC, Acrylic, Polyethylene, Polypropylene and PTFE Fluoropolymer (Teflon®)
High Surface Energy (HSE)
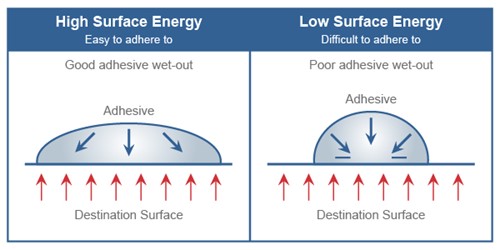
The higher the surface energy of the application material, the easier the label will stick. Typical high surface materials include: Copper, Stainless Steel, Aluminum, Zinc, Tin, Glass, Nylon, Polyester (PET), ABS Plastic, Polycarbonate. To gain the best possible adhesion, an adhesive must thoroughly “wet out” the surface to be bonded.
What does wetting out mean?
“Wetting out” means the adhesive flows and covers a surface to maximize the contact area and the attractive forces between the adhesive and bonding surface.
See image below…
A good example to explain surface energy is how water behaves when dropped onto the surface of waxed and un-waxed cars. Consider water dropping on an un-waxed car bonnet. The surface energy here is high and the water visually flows into puddles. Now consider a bonnet which has been waxed, the surface energy is now low and the water forms into beads rather than flowing out.